열가소성 복합재료(TPC)는 기존 열경화성 복합재료에 비해 빠른 가공 속도와 여러 번 재가열 및 재성형이 가능하다는 등 여러 가지 장점을 제공합니다. 또한, 용융 및 응고 과정이 화학 반응이 아닌 물리적 변화를 수반하기 때문에 재활용이 가능하고 제조 효율성을 향상시킬 수 있습니다.
하지만 최적의 성능을 얻으려면 열처리 공정을 정밀하게 제어해야 합니다. 이 공정은 가열, 용융 가공 및 냉각 단계를 포함하며, 특히 냉각 과정에서 고분자가 원하는 상태에 도달하도록 세심한 주의를 기울여야 합니다.
열경화성 복합재료의 경우 가열하면 점도가 감소하고 겔화가 촉진되는 것과 달리, TPC에서는 냉각 단계가 결정화도와 최종 물성을 제어하는 데 가장 중요합니다.
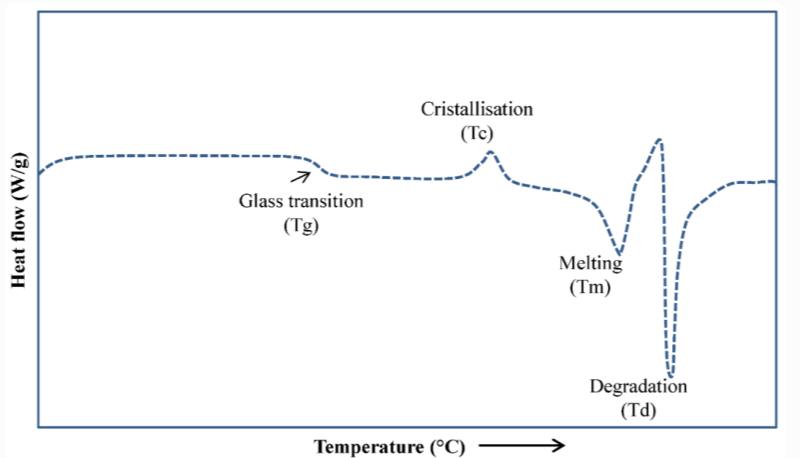
고분자 매트릭스의 열적 거동을 이해하는 것은 효과적인 열처리에 필수적입니다. 재료 공급업체는 일반적으로 다음과 같은 주요 매개변수를 제공합니다.
이러한 특성은 일반적으로 온도의 함수로서 열 흐름을 추적하는 시차 주사 열량계(DSC)를 사용하여 측정됩니다.
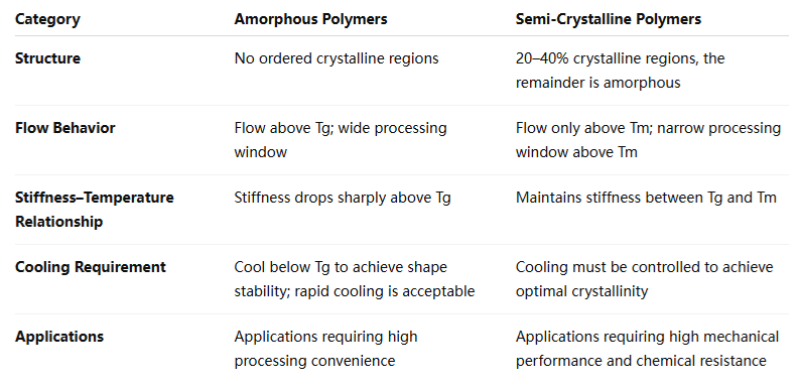
비정질 고분자와 반결정질 고분자 중 어떤 것을 선택하느냐에 따라 열처리 요구사항이 크게 달라집니다.
비정질 고분자 규칙적인 결정 구조가 부족하고 온도가 유리전이온도(Tg)를 초과하면 유동성을 나타내기 시작합니다. 온도가 증가함에 따라 점도가 점차 감소하여 비교적 넓은 가공 범위를 제공합니다.
반결정성 고분자 일반적으로 20~40%의 결정성을 포함합니다. 유리전이온도(Tg)와 극한온도(Tm) 사이에서는 강성을 유지하며, Tm에 도달한 후에야 유동성이 나타나기 시작합니다. 따라서 가공 범위는 좁지만 고온 성능은 우수합니다.
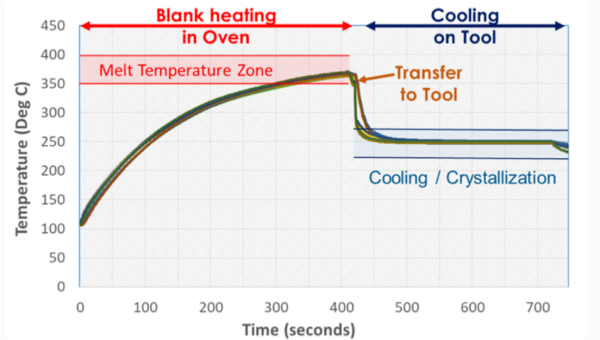
가열 단계에서는 TPC를 가공 온도(Tp)까지 가열하여 열분해 없이 성형에 필요한 충분히 낮은 점도를 얻습니다.
비정질 고분자의 경우 전이는 Tg에서 발생하고, 반결정성 고분자의 경우 Tm에서 발생합니다. 제품 데이터시트에는 일반적으로 권장 가공 온도 범위가 명시되어 있습니다.
PAEK 계열과 같은 고성능 폴리머의 경우, 가공 중 산화를 방지하기 위해 상한 온도(일반적으로 약 400°C)가 정해져 있습니다.
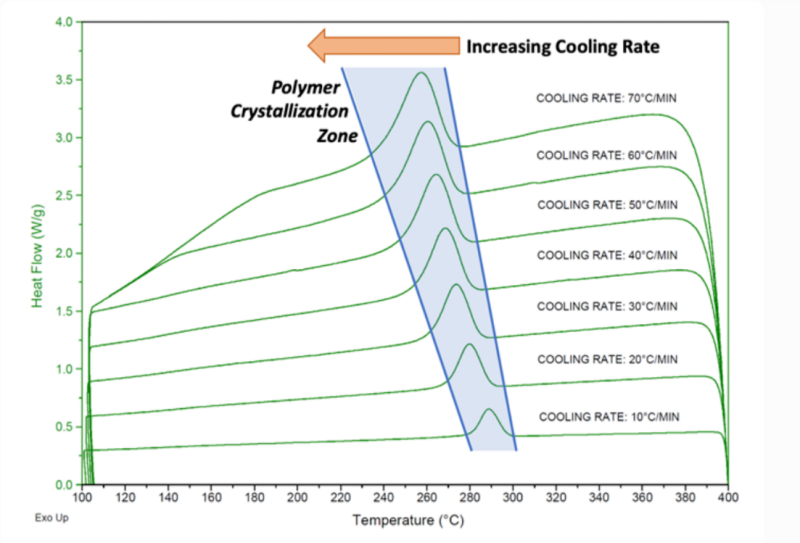
냉각은 열가소성 복합재료, 특히 반결정성 고분자에 있어 가장 중요한 단계입니다. 냉각 과정이 결정성과 최종 성능을 좌우하기 때문입니다.
비정질 TPC는 치수 안정성을 얻기 위해 유리전이온도(Tg) 이하로 냉각만 하면 되므로 매우 빠른 생산 주기가 가능합니다.
반결정성 고분자는 핵 생성 및 결정 성장을 위해 유리전이 온도(Tc) 범위 내에서 제어된 냉각이 필요합니다.
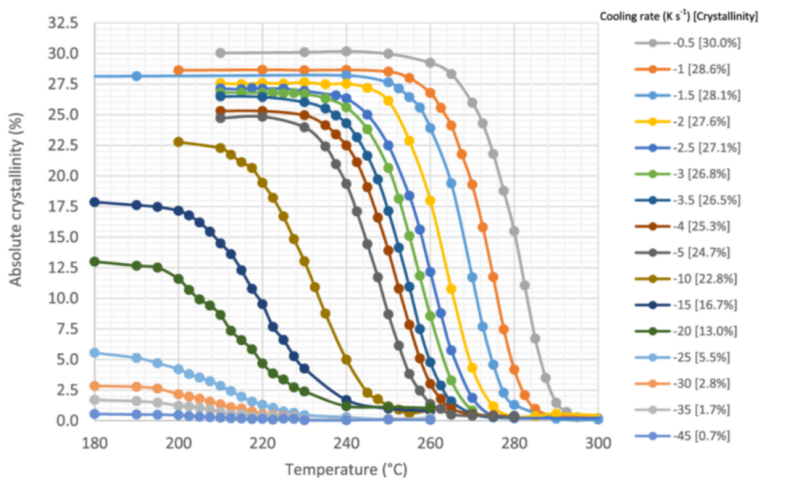
결정성은 환경 저항성과 기계적 특성에 큰 영향을 미칩니다. 최적의 결정성 수준은 일반적으로 20%에서 40% 사이입니다.
냉각 속도가 높을수록 Tc가 낮아지고 결정화 범위가 넓어지지만, 지나치게 빠른 냉각은 불완전한 결정화를 초래할 수 있습니다.
냉각은 반결정성 고분자의 부피 변화를 일으켜 내부 응력을 발생시킵니다. 예측 모델링 도구를 사용하면 엔지니어는 최적화된 금형 설계를 통해 이러한 영향을 보정할 수 있습니다.
열가소성 복합재료는 적층, 압축, 성형 및 용접과 같은 여러 열 사이클을 견딜 수 있습니다. 적절하게 제어될 경우, 고성능 열가소성 수지는 Tm 이상으로 반복 가열된 후에도 안정적인 기계적 특성을 유지합니다.
열처리는 열가소성 복합재의 성능을 최적화하는 데 중요한 역할을 합니다. 냉각은 반결정성 시스템에서 결정화도와 치수 안정성을 제어하는 반면, 비정질 고분자는 제조 공정을 단축할 수 있도록 합니다.
적절한 열 관리를 통해 제조업체는 항공우주 및 자동차와 같은 까다로운 산업 분야에 필요한 고성능 부품을 안정적으로 생산할 수 있습니다.